VIBRANT SOUNDBRIDGE VORP 503
Active Middle Ear Implant

Outstanding Hearing, Proven Reliability
VIBRANT SOUNDBRIDGE (VORP 503) is a unique active middle ear implant that combines excellent hearing outcomes with flexible surgical handling and proven long-term reliability. It is an ideal option for patients five years or older with:
- Mild-to-severe sensorineural hearing loss resulting from inner ear pathologies when acoustic hearing aids are contraindicated.
- Mixed hearing loss resulting from middle ear pathologies, including conductive hearing loss.
- Excellent hearing outcomes
- Straightforward surgery
- Proven long-term reliability

Hearing Performance
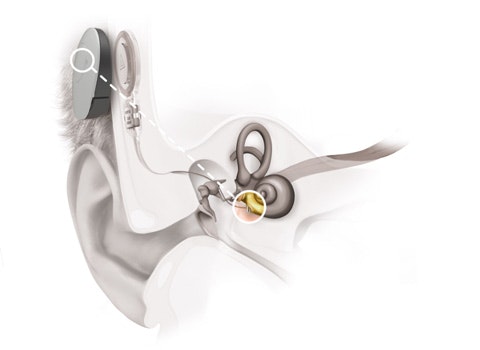
SOUNDBRIDGE provides recipients with natural sound quality without feedback. That’s thanks to its unique design with a Floating Mass Transducer (FMT) that can be coupled to the most appropriate middle ear structure to suit each individual recipient’s specific anatomy.
For patients with sensorineural hearing loss, the FMT is attached to the incus, allowing stimulation of the inner ear via the ossicles, making hearing with SOUNDBRIDGE very similar to hearing naturally. In 127 studies with over 1,600 patients, SOUNDBRIDGE has shown to be an effective treatment for sensorineural hearing loss in its indication criteria. Combined data from these studies show improvements from 43.6% to 78.6% in word recognition scores at 65dB SPL for patients with sensorineural hearing loss. [ft]
- Powerful and focused
- True binaural hearing
- Natural sound quality
- Feedback-free
For patients with mixed hearing loss, SOUNDBRIDGE bypasses middle ear issues and directly stimulates vibratory ear structures. SOUNDBRIDGE can deliver great speech understanding in noise, natural hearing quality, and excellent sound localization. [ft],[ft] Hearing improvements with SOUNDBRIDGE remain stable in the long term. [ft]
Straightforward Surgery
Standardized single-point FMT attachment and self-drilling screws make SOUNDBRIDGE implantation fast and straightforward—while maximizing surgical flexibility. SOUNDBRIDGE is suitable for implantation for recipients age five or older because the FMT is attached at a single point, making it independent from skull growth. In fact, it is the only middle ear implant on the market featuring single-point attachment.
In addition, the SAMBA 2 audio processor for SOUNDBRIDGE combines intelligent hearing technology with easy handling, a variety of connectivity options, and intuitive fitting software to make hearing as simple as possible—for both hearing implant recipients and professionals.
- Standardized coupling
- Fast and stable fixation
- Surgical flexibility

Peace of Mind
SOUNDBRIDGE provides stable audiological results in the long term with an exceptionally low rate of complications. Thanks to the intact skin design, SOUNDBRIDGE avoids issues such as infections and additional clinic visits common with BAHA devices. In addition, recipients and their radiologists get peace of mind knowing that the VORP 503 may be safely MRI scanned at 1.5 Tesla.*

- Long-term reliability
- Exceptionally low complication rate
- MRIs at 1.5 Tesla
MR Conditional at 1.5 Tesla
The VORP 503 magnet was designed with MRI scans in mind. This allows the implant to be exposed to MRI scans at 1.5 Tesla without magnet removal.* Because of our long and positive experience with MRIs and hearing implants, we offer a life-long MRI guarantee. In the very unlikely event that it’s damaged during an MRI scan we will replace the implant.**
For the patient, this means no surgery is necessary for an MRI scan, and of course, there is no hearing downtime—patients can use the implant immediately after scans.***

Why MED-EL:
A Trusted Partner
For more than 35 years, MED-EL has been a trusted partner and innovation leader in hearing implants. We’re here to work together with you, and we’re committed to providing outstanding service and support for our professional partners.
With the most advanced cochlear implant systems, we offer the best hearing experience for your patients and the best clinical experience for you.
- Closest to Natural Hearing
- Superior Ease-of-Use
- True Peace of Mind
Contact Us
Ready to learn more about VIBRANT SOUNDBRIDGE?
Fill out our simple contact form and we’ll get in touch with you.
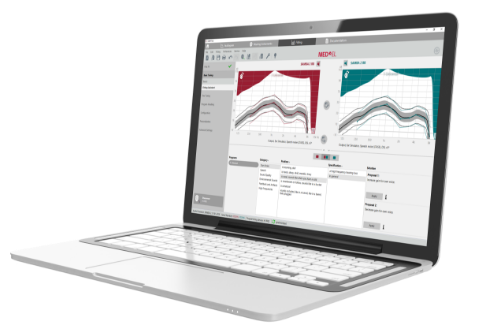
SYMFIT 8.0
The SYMFIT 8.0 software makes fitting SAMBA 2 easy and efficient. Its straightforward guided workflow includes precise Vibrograms and fitting tools to help you get the most out of SAMBA 2.

SAMBA 2
SAMBA 2 for VIBRANT SOUNDBRIDGE and BONEBRIDGE combines intelligent hearing technology with easy handling, intuitive connectivity options and improved fitting software to make hearing as simple as possible—for both hearing implant recipients and their audiologists.
Technical Data


VORP 503 Active Middle Ear Implant
VIBRANT SOUNDBRIDGE System
Contents of Implant Kit
- 1 VORP 503 middle ear implant
- 3 self-drilling cortical screws
- 1 single-use screwdriver
Weight
- 10.6 g
Materials in Contact With Tissue
Implant: medical grade silicone elastomers, titanium, medical grade epoxy Screws: titanium alloy Ti 6Al 7Nb
Recommended Bone Bed Depth for Demodulator
1.9 mm
Safety Features
Overvoltage protection
MRI Conditions
MR-conditional at 1.5 Tesla*
Screw Penetration in Bone
3.5 mm (max.)
Maximum Immersion Depth
50 m in salt water (6 bar)
Biocompatible in Accordance With ISO 10993-1
Latex-free****
Delivered sterile
Not for Reuse
Vibroplasty Couplers
Incus-SP-Coupler
- For placement onto the short process of the incus
- Material in contact with tissue: titanium grade 5 ELI (ASTM F136)
- Contents of kit: 1 Coupler, 1 holding frame, 1 retainer
Incus-LP-Coupler
- For placement onto the long process of the incus
- Material in contact with tissue: titanium grade 5 ELI (ASTM F136), titanium grade 2 (ASTM F67)
- Contents of kit: 1 Coupler, 1 holding frame, 1 retainer
RW-Soft-Coupler
- For placement onto the round window membrane
- Material in contact with tissue: Medical silicone and silicone gel
- Contents of kit: 2 Couplers, 1 holding frame, 2 retainers
Vibroplasty-CliP-Coupler
- For placement onto the stapes head
- Material in contact with tissue: titanium grade 2, titanium grade 1
- Contents of kit: 1 Coupler
Stapes-SH-Coupler
- For placement onto the stapes head
- Material in contact with tissue: Titanium Grade 2 (ASTM F67) and Titanium Grade 5 (ASTM F136)
- Contents of kit: 1 coupler, 1 holding frame, 1 retainer
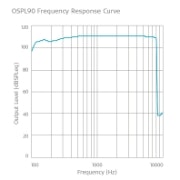
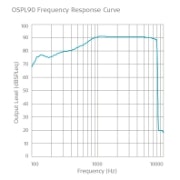
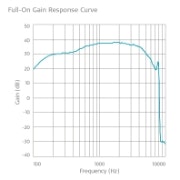
Frequency Response Curves
SAMBA 2 Hi
Maximum OSPL90: 109 dB SPLeq
HFA-OSPL90: 109 dB SPLeq
Maximum full-on gain: 53 dB
HFA-FOG: 52 dB
Equivalent input noise: 23 dB SPL
Total harmonic distortion, 70 dB SPL input level, 500 Hz distortion test frequency: < 5 %
Total harmonic distortion, 70 dB SPL input level, 800 Hz distortion test frequency: < 5 %
Total harmonic distortion, 65 dB SPL input level, 1600 Hz distortion test frequency: < 3 %
Total harmonic distortion, 60 dB SPL input level,
3200 Hz distortion test frequency: < 3 %
SAMBA 2 Lo
* The SOUNDBRIDGE VORP 503 implant is MR conditional. Recipients with the SOUNDBRIDGE VORP 503 implant may be safely MRI scanned
at 1.5 Tesla following the conditions detailed in the instructions for use.
The previous generations, VORP 502 and VORP 501, are MR unsafe and should not be scanned.
** Provided that the MRI is conducted according to the IFU or the medical procedures manuals.
*** No surgery unless required for diagnostic reasons.
**** Where “latex-free” means “not made with latex” in accordance with the latest FDA “Recommendations for Labeling Medical Products
to Inform Users that the Product or Product Container is not Made with Natural Rubber Latex” guideline, 2014
Please find detailed information and specific conditions for use for each product in the respective instructions for use.