360° View of Cochlear
Implant Safety and Reliability

Our Philosophy
Hearing connects us to so many of life’s cherished moments. That’s why we’re always here to help your patients with hearing loss experience these wonderful moments day after day.
Since the very beginning, the development of our life-changing devices has been guided by our unique 360-degree view of reliability and safety. It’s not only about making our cochlear implants and audio processors as reliable as possible. Electrode design, safe stimulation and MRI safety are also integral aspects of cochlear implant safety. Without taking these into account, creating a safe and reliable hearing experience just isn’t possible.
Every MED-EL recipient is a person who trusts us. We know they count on us to connect them to everything they love every day, and we are committed to providing them with devices that meet the highest quality, safety, and reliability standards on the market.

Leading in Cochlear Implant Reliability
All of our latest-generation titanium implants have an overall cumulative survival rate over 99%—reliability performance that no other cochlear implant manufacturer has achieved. [ft][ft][ft]
The outstanding reliability of our cochlear implants is not by chance. It is the result of the focused oversight and expert craftsmanship that takes place in our state-of-the-art manufacturing facilities. Unlike other cochlear implant producers who outsource manufacturing overseas, every single MED-EL implant is produced at our global headquarters in Austria—and it has been that way for over 35 years. This allows us to consistently maintain the highest standards of European quality and engineering.
We take every detail into consideration, from quality-oriented design and development to vigorous testing and clinical support. With our cutting-edge processes, superior components, and exceptional quality control, it is no coincidence that every MED-EL cochlear implant meets the highest standards for quality, safety, and reliability. But don’t take our word for it—the data speaks for itself.
- 360° comprehensive approach to safety & reliability
- All implants manufactured in Austria
- Unmatched backwards compatibility
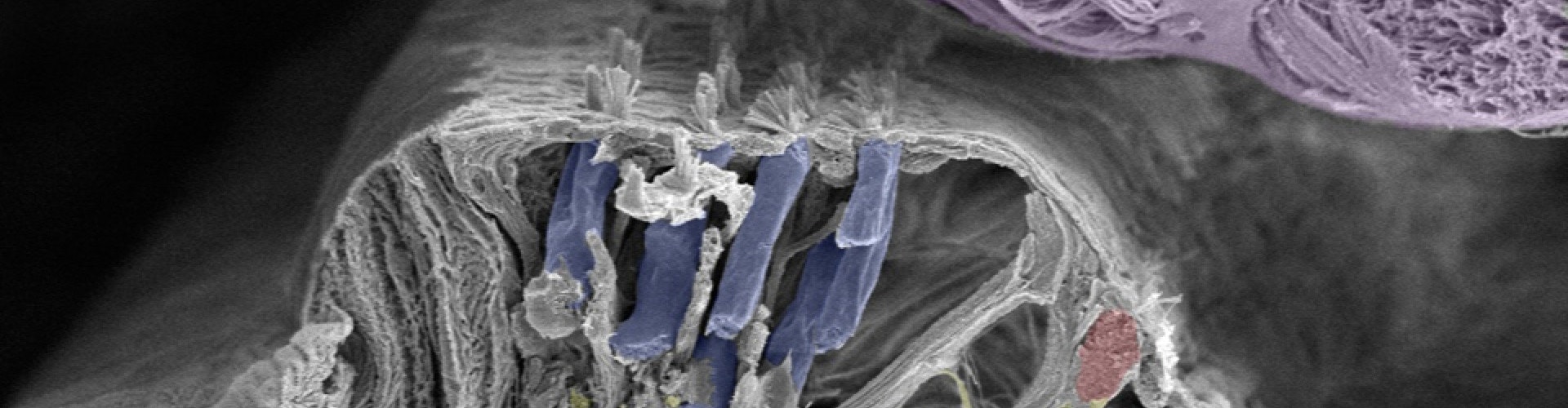
Electrode Safety: For Atraumatic, Deep Insertion
A deaf ear is not a dead ear, and the cochlea is filled with intricate structures that we want to protect. That’s why, for more than 35 years, we’ve worked to create our ultra-flexible electrode arrays. Uniquely engineered, our electrode arrays are the most atraumatic electrode arrays available.[ft] They reduce the chances of tip fold-over, preserve the delicate structures of the inner ear, and gently adapt to the individual cochlea for deep, atraumatic, and reliable electrode insertion.
- Thinnest full-length electrode array
- Safe and reliable placement
- No harm to basilar membrane

Safe Stimulation
When it comes to the long-term safety of cochlear implants, safe stimulation is an absolute must. If direct current leaks into the cochlea, it could result in the dissolution of electrode contacts or even damage to neural tissue. [ft][ft][ft][ft][ft][ft][ft][ft] That’s where safety capacitors come in. They work as a gatekeeper, letting alternative current flow back and forth, while blocking harmful direct current, ensuring safe stimulation.

Independent Safety Capacitors
Unlike some cochlear implant manufacturers, MED-EL has an independent safety capacitor for every electrode channel. That way, we can ensure safe, precise, and fast stimulation in the long term. [ft][ft]

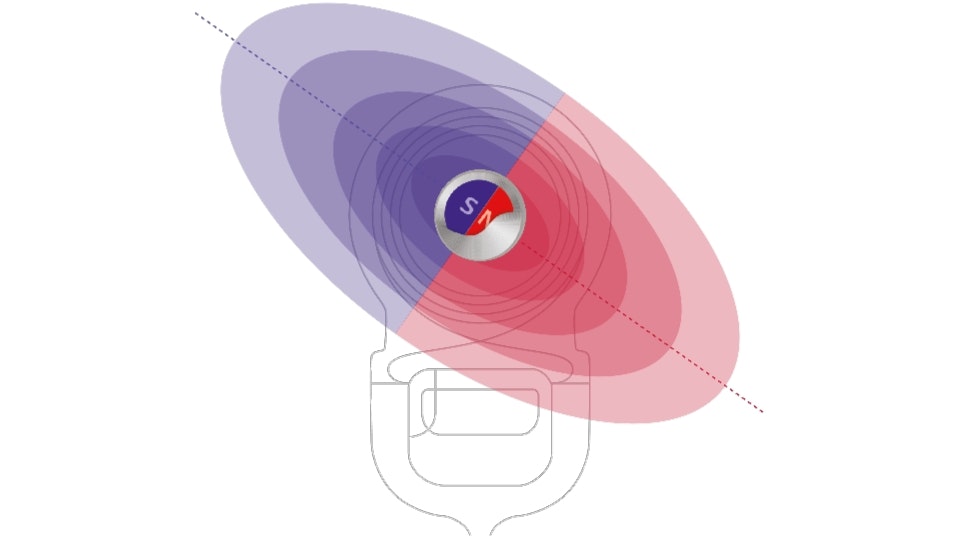
Made for MRI
Creating a reliable cochlear implant means creating one that’s ready for the future. With most of your patients needing an MRI in the next 10 years, we believe that every single hearing implant should be designed for outstanding MRI safety.** That’s why we created the revolutionary SYNCHRONY implant magnet—a rotatable, self-aligning magnet that enables 3.0 Tesla MRI scans** with no surgery,*** and no hearing downtime.
Great Protection. Guaranteed.
Because of our long and positive experience with MRIs and cochlear implants, we offer a life-long MRI guarantee. In the very unlikely event that it’s damaged during an MRI scan we will replace the implant.**** Our MRI guarantee is:
- Valid for all MED-EL multichannel cochlear implants since 1994.
- Life-long and worldwide.
- The first and only offered by any hearing implant producer.
With no need to remove the magnet, and a guarantee against damage during an MRI we can offer your patients a reliable cochlear implant experience whatever life brings.

Audio Processor Reliability
It’s not just our implants that are designed to deliver outstanding reliability, but our audio processors too. With a reliable audio processor, your patients save themselves time, money, and hassle. And it’s less trouble for hearing professionals: With fewer issues, you’ll save time on fitting replacements and administrating returns.

MED-EL:
A Reliable Partner
For more than 35 years, MED-EL has been a trusted partner and innovation leader in hearing implants. We create and manufacture our implants directly in our Austrian headquarters, ensuring they follow state-of-the-art European engineering, and meet the highest standards in quality and reliability. With this focused oversight and our dedication to quality, we can check and verify every single step of the design and process.
MED-EL has always been privately owned by our founders, so for us reliability is not a percentage or an earnings report for investors. With no shareholders to answer to, we’re in a unique position where we can put the safety and peace of mind of our recipients above all else. And because we believe that everyone should benefit from the latest hearing technology, our latest audio processors work for all our recipients since 1994.
Choosing MED-EL means putting your trust in us. We promise to always be there for you, our professional partner, and for your patients.
- European engineering
- Manufactured on-site
- Dedication to reliability and quality
- Unmatched backwards compatibility
- Committed to recipients
Contact Us
Want to learn more about MED-EL and our life-changing hearing solutions? Simply fill in the form below.
* All registered and currently marketed implants are included in the reliability calculations, and every explanted and returned implant is subject to systematic failure analysis. All failures are classified and Cumulative Survival Rates (CSR) are calculated
in accordance with ISO 5841-2:2014. All confirmed device malfunctions including accident-related failures are considered for reporting. Failures traced back to induced malfunctions are not included in the reported data in compliance with the ISO standard.
The results of the calculations are reported following principles of the European Consensus on Cochlear Implant Failures and Explantations, with adults and children being shown separately and with 95% confidence intervals. Please be aware that confidence
intervals smaller than 0.1% may not be clearly visible in the graphs.
The sample size of each model and population are not provided. MED-EL publishes reliability data one year after the first implantation at the earliest.
** MED-EL
cochlear implants since 1994 are MR conditional. Recipients with a MED-EL cochlear implant may be safely MRI scanned following the conditions detailed in the instructions for use.
*** Unless required for diagnostic reasons.
**** The terms
and conditions of the MRI Guarantee can be found here.